Native Orthopaedics Receives additional FDA 510(k) Clearances for the Repair Construct Across Shoulder, Foot and Ankle, and Knee Indications
PROVIDENCE, RI
Native Orthopaedics has received FDA 510(k) clearance for the Repair Construct — a knotless, intraoperatively adjustable suture fixation product powered by the company's two proprietary technologies - Dragonfly™ finger trap and Zerofly™ integrated soft button fixation.
The cleared indications include soft tissue-to-bone fixation in the shoulder (biceps tenodesis, AC separation repair), foot and ankle (Achilles tendon repair), and knee (meniscal root repair, MPFL repair, ACL/PCL repair). Native’s initial commercial focus will be on the many types of ACL and PCL repair.
“We introduced our technologies to surgeons that address meniscal root injuries, because this market has been drastically underserved and the existing solutions were challenging to use and lead to sub-optimal outcomes. Our platform technologies address the shortcomings of all the other systems on the market, by uniquely addressing construct creep, imprecise tensioning, and providing the ability to dynamically adjust the tissue tension intra-operatively.
Surgeons that have used our meniscal root offering, not only thank us but also see the clinical benefits of our platform for multiple use cases in their daily surgery schedules. We are proud to see this 2nd additional FDA clearance unlock our ability to serve these surgeons and many others in the future for additional soft tissue injuries. ”
How the Technology Works
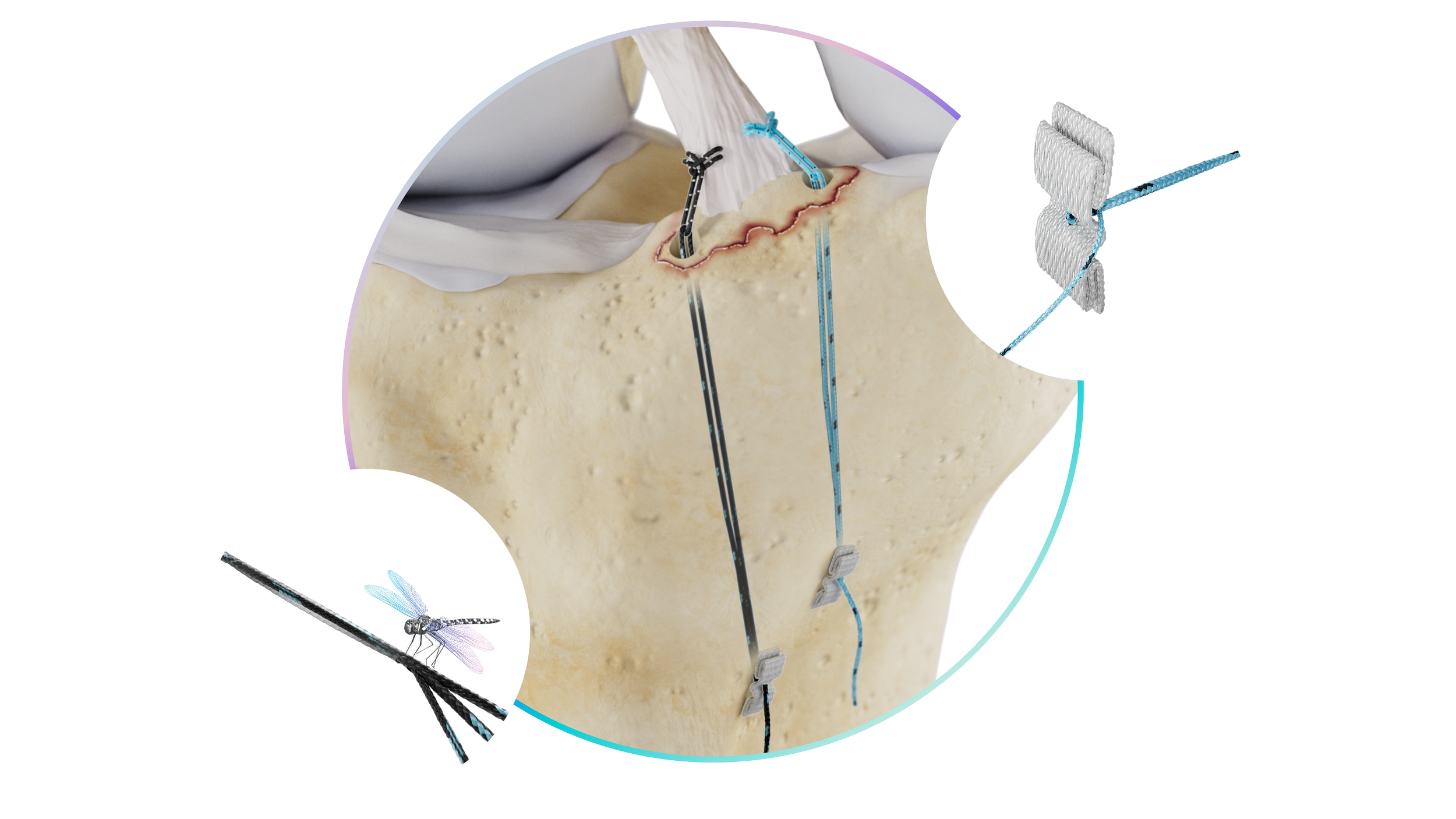
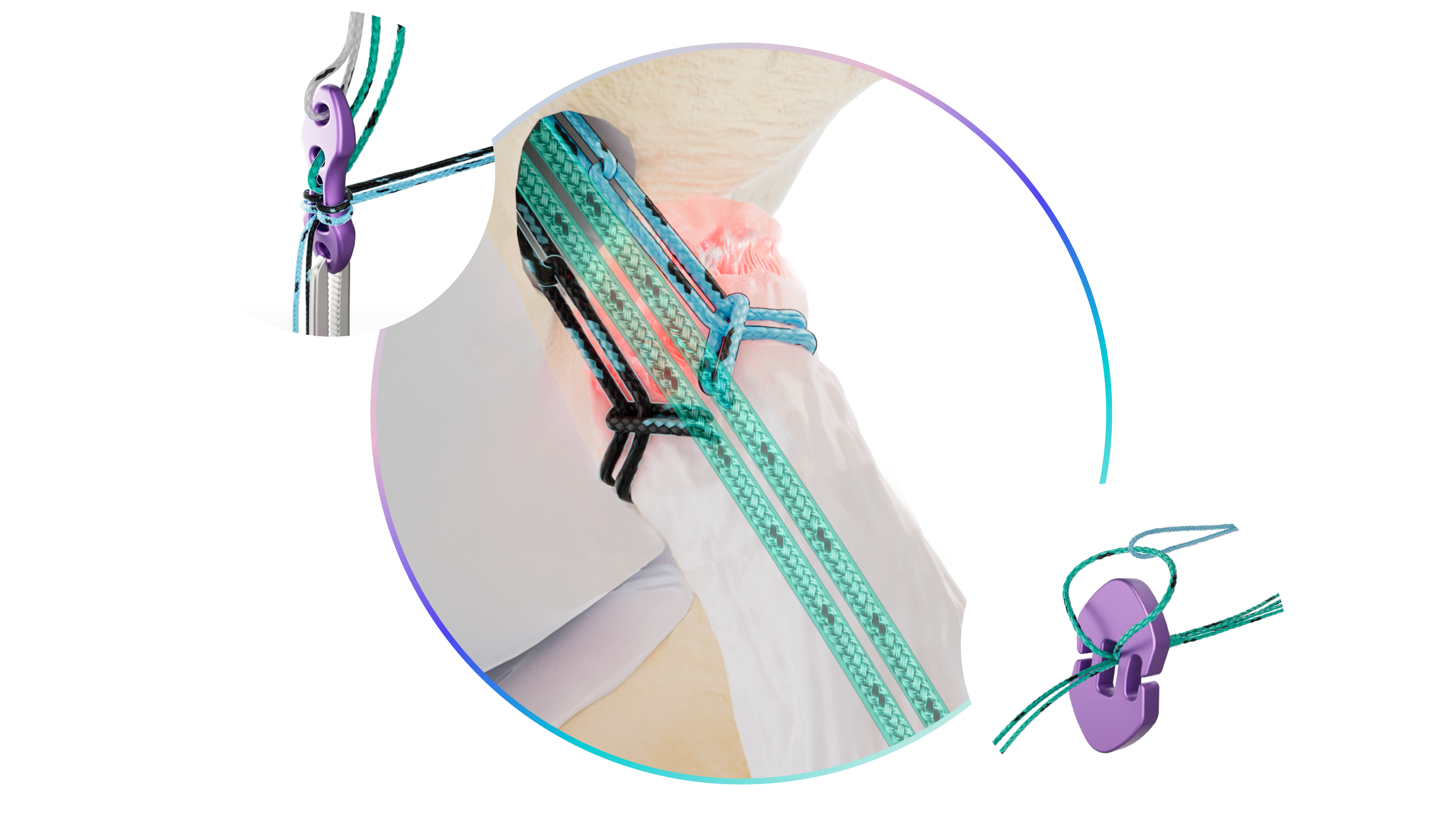
Each Repair Construct is powered by a Dragonfly quad core advanced finger trap, for simple, independent tissue tensioning. The surgeon securely attaches the Repair Construct to the tissue, shuttles the construct through a bone tunnel, and connects it to Native's titanium Bow-Tie button, or can select a Repair Construct that is preassembled with the patented Zerofly soft button fixation. Zerofly is a metal free cortical fixation technology designed to conform to the patient’s anatomy, while providing versatility for the surgeon. Once shuttled through the tunnel, simply pull the tail of the Dragonfly to optimize the tension of the construct and reduce the tissue to the bone.
After initial fixation, the surgeon can cycle the joint and pull the tail again to remove additional laxity that may develop from viscoelastic creep. Each construct can be re-tensioned independently without disturbing the rest of the repair. Most fixation systems don't allow this.
Dragonfly's quad-core architecture — a finger trap within a finger trap — doesn't rely on a secondary locking feature to prevent slippage. The construct is protected by 21 issued patents, with 30+ additional applications pending.
Additionally, Native received a third FDA 510(k) clearance for the Native Bridge - a novel construct intended to be used for fixation of bone to bone, and is intended as fixation posts, a distribution bridge, or for distributing suture tension over areas of ligament or tendon repair in ACL/PCL repair.
-
Native Orthopaedics is a company built by a Surgeon for Surgeons. We are innovators and doers who want to help those with knee pain live life to its fullest. We're committed to crafting creative, end-to-end surgical solutions and innovative tools that push the field of orthopaedics into the future with simpler, more effective solutions that preserve our native anatomy. Native Orthopaedics is a privately held company actively fundraising, with a series A round anticipated in the coming months. Learn more at www.nativeortho.com
-
Brian Morley, Chief Marketing Officer
bmorley@nativeortho.com -
Tom Gamache, CEO & Co-Founder
tgamache@nativeortho.com -
1. Data on file with Native Orthopaedics, Inc.
Rx only. Caution: Federal law restricts this device to sale by or on the order of a physician. Refer to package insert for indications, contraindications, warnings, precautions, and instructions for use.